International Journal of Anatomical Sciences 2013, 4(2):14-19
Research Paper
Coronary Venous Anatomy and its Relevance
Suma HY, Hariharan DR, Ramachandra Rao K.
Department of Anatomy, Jawaharlal Institute of Postgraduate Education and Research, Puducherry, India.
Department of Anatomy, Saveetha Medical College, Chennai.
Key words: coronary sinus, cardioplegia, thebesian valve, cardiac resynchronization therapy
Abstract: Coronary veins are not given its due importance unlike the arteries. It is being increasingly used in cardiothoracic surgeries and electrophysiological studies. Hence, the present study has been taken up to know the pattern of coronary venous anatomy and its relevance in the clinical field. The coronary sinus and its tributaries were studied by anatomical dissection in 25 adult human cadaveric hearts, which had been fixed in formalin solution. Coronary sinus was studied in relation to its location, length, and arteries accompanying it, tributaries and their variations. From the present study it has been found that the length of coronary sinus varies in between 2 cm to 5.5 cm. It was always located in the coronary sulcus, to the left of crus. It was accompanied by circumflex artery in half of the cases and also by right coronary in few. One specimen had middle cardiac vein directly opening into the right atrium. The great cardiac vein and middle cardiac vein was present in all specimens. In 64% of the hearts thebesian valve was membranous and crescentic. In 20% the ostium was valve less. This study highlights the importance of coronary venous anatomy. It is of relevance in catheterization of the coronary sinus for various procedures like in cardioplegia and electrophysiology.
Variations pertaining to the coronary arterial system have been described but less attention has been paid to the venous system. Coronary veins are not affected by atherosclerosis. In hearts affected by arteriosclerosis of arterial system, there is a richer, more uniform venous network in the subendocardial zone (Permyos et al., 2001).
The cardiac venous system, has many applications in invasive cardiology which are 1) targeted drug delivery 2) retrograde cardioplegia administration 3) providing potential conduits to bypass coronary artery stenosis and 4) to deliver stem cells to the infracted myocardium (Singh et al., 2005).
Coronary sinus is utilized to access the left atrial and left ventricular epicardium. Diagnostic and mapping maneuvers can be done through this route to determine the type of arrhythmia and also permit the delivery of ablative energy (Cappato et al., 1997).
For cardiac resynchronization therapy in patients with heart failure, endocardial implantation will be successful if the left ventricular lead can be positioned in a vein draining that region with the latest mechanical activation (Nico et al., 2006). Failure of left ventricular lead placement has been attributed to the inability to insert catheter in the coronary sinus and the lack of suitable side branches (Abraham 2003, Puglisi et al., 2004).
The venous system is a useful conduit for delivery of percutaneous transcatheter treatment. Retrograde cardioplegia is achieved through the cardiac veins during cardiac surgeries and may help to flush the distal emboli from the arterial system (Iyer Praveen B, 2006).
Hence the present study was undertaken to know the coronary venous anatomy in cadaveric hearts in this region.
Materials and Methods
The coronary sinus and its tributaries were studied by anatomical dissection in 25 adult human cadaveric hearts, which had been fixed in formalin solution. Coronary sinus was studied in relation to its location, length, and arteries accompanying it. Length of the coronary sinus was measured from the point where oblique vein of left atrium opens the coronary sinus. Second point was taken at the junction, where coronary sinus pierces the wall of the right atrium.
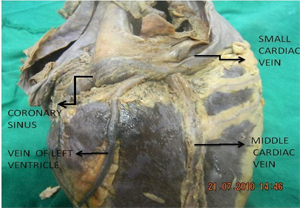
The tributaries were also studied by careful dissection on the epicardial surface. Small, great, posterior, and middle cardiac vein were the first order tributaries, as they are made out well during routine dissection. Hence these have been mentioned in the present study. But the second and third order tributaries were difficult to dissect.
We measured the cranio caudal and transverse dimensions of the coronary sinus ostium and looked for the shape of the ostium, morphology of the thebesian valve. All the measurements were done using vernier caliper
Table 1 Coronary veins in relation to various studies
| Ortale J 2001 | Gerber TC 2001 | Longbloed 2005 | Present study 2012 | ||
| Great cardiac vein | 100% | >90% | 100% | 100% | |
| Middle cardiac vein | 100% | >90% | 100% | 100% | |
| Small cardiac vein | 54% | * | 46% | 64% | |
| Left marginal vein | 97% | <50% | 61% | 92% | |
| Vein of left ventricle | * | <50% | 95% | 92% | |
| Anterior cardiac vein | * | * | * | 16% | |
| Oblique vein of left atrium | * | * | 34% | 36% | |
* Percentages of that tributary was not mentioned in that particular study
Table 2 Diameters of the coronary sinus ostium
| With valve (in mm) | Without valve (in mm) | |||||
| Present study 2012 | Gary S Mak 2009 | Present study 2012 | Gary S Mak 2009 | Jongbloed 2005 | ||
| Transverse diameter | 5.04 | 7.3+2.8 | 10.11 | 9.4+2.9 | 14.7+4.4 | |
| Cranio caudal diameter | 6 | 7.9+2.7 | 7.44 | 9.3+2.9 | 11.9+3.5 | |
Table 3 Thebesian valve morphology
| Morphological characters | Herman et al. | Present study | ||
| 1 | Valveless | 14.7% | 20% | |
| 2 | Crescentic, membranous | 38% | 46% | |
| 3 | With fenestrations | 5.3% | 6% | |
| 3 | Common Eustachian and thebesian valve | 6% | 8% | |
| 3 | In some- completely occluding the ostium | 30.7% | 20% | |
Table 4 Arteries accompanying coronary sinus
| NAME OF ARTERY | Incidence | ||||
| OUT OF 25 | % | ||||
| Circumflex Artery | 13/25 | 52% | |||
| Right Coonary Artery | 5/25 | ||||
| Not accompanied by any vessel | 5/25 | 20% | |||
| Both Vessels | 1/25 | 4% | |||
Length of the Coronary sinus
The coronary sinus has been reported to be as long as 5.4 cm but is usually about 2.5 cm in length(Susan Standring, 2008). In the present study the length of coronary sinus varied from 2 to 5.5cm. Oblique vein of left atrium was observed in very few cases. Duct of cuvier was absent in the all the specimens. Middle cardiac vein directly opened into right atrium in one case. In a few, coronary sinus was intramural i.e. covered by few myocardial fibers.
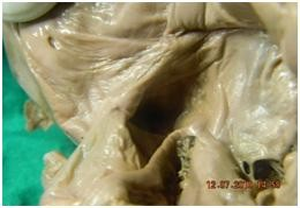
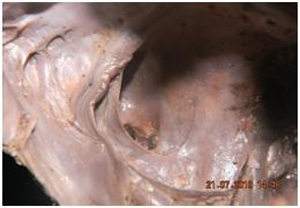
| Fig 1 Coronary sinus opening with a small muscular valve | Fig 2 Coronary sinus opening with crescentic valve |
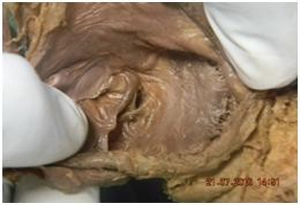
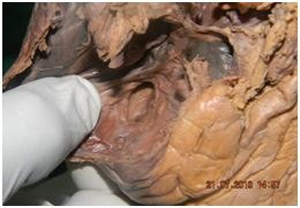
| Fig 3 Coronary sinus opening with membra-nous valve | Fig 4 Coronary sinus opening without valve |
| Fig 5 Location of great cardiac vein in the anterior interventricular groove | Fig 6 Tributaries of coronary sinus |
Discussion
There are three venous systems that drain the heart: the coronary sinus, the anterior cardiac veins, and the venae cordis minimae. The coronary sinus receives all cardiac veins except venae cordis minimae and anterior cardiac veins. The coronary sinus opens into the right atrium between the opening of the inferior vena cava and the tricuspid orifice. It returns blood to the right atrium from nearly all regions of the heart, including the septa, and accounts for 75% of the coronary venous circulation. Studies to categorize variations in cardiac venous circulation based on the regions drained by cardiac veins have not produced any accepted pattern (Susan Standring, 2008).
According to the direction of flow of blood, two major variants can be distinguished: a majority (70%) in which the small cardiac vein is independent, small or absent. And a less frequent pattern (30%) in which this vein connects both coronary and anterior cardiac systems (Susan Standring, 2008).
Coronary sinus may be obliterated or absent. The great cardiac vein then drains into the superior venacava or left brachiocephalic vein via the right superior venacava. Several veins, including the middle cardiac vein, may converge to empty into a common opening into the right atrium. Coronary sinus may open into left atrium (Ronald et al., 2010). In the present study coronary sinus was present in all the cases.
Variations of the coronary veins, may cause non homogenous distribution of retrograde cardioplegia and result in inadequate myocardial protection. In 1987, Ludinghausen examined 350 hearts and defined five groups (Types A, B, C, D & E) of cardiac vein variations (Permyos et al., 2001).
Type A anterior and small cardiac veins enter the right atrium
Type B all major veins enter the coronary sinus
Type C anterior cardiac veins enter the right atrium
Type D great cardiac vein and vein of left ventricle enter the coronary sinus.
Type E Only veins of the left ventricle enter the coronary sinus.
In the present study the cardiac veins belonged to Type A in majority of cases (64%). Remaining hearts belonged to Type B (22%). Only in one case(4%) middle cardiac vein opened directly into right atrium.
Types D & E causes non-homogenous distribution of cardioplegia, hence inadequate myocardial protection.In some cases left ventricle and interventricular septum are poorly perfused due to inadequate drainage by the veins i.e. middle and small cardiac veins (Type D & E).
Great and middle cardiac vein was present in all cases as observed by previous authors. Even in the present study these veins were present in all the specimens (Table 1). But left marginal and vein of left ventricle are present in 50-75% of the heart (Ortale et al., 2001; Gerber et al., 2001; Morique et al., 2005). In the present study, these veins were present in 92%. This can be explained because; those cases where left marginal vein is absent had a previous history of myocardial infarction. This may limit optimal left ventricular lead positioning for cardiac resynchronization therapy (Nico et al., 2006).
It has been observed that richer, more uniform venous network exists in the subendocardial zone of both ventricles in those with heart disease (Permyos Ruengsakulrachann et al., 2001). Hence cardiac veins are being preferred for cardioplegia. Not only the lack of suitable side branches but also obstruction and ostial narrowing (e.g., by a thebesian valve) may complicate left ventricular lead implantation (Monique et al., 2005)
Thebesian valve morphology (Table 2 & 3)
Coronary sinus opening is guarded by Thebesian valve. Six types of variations were observed involving valve of the coronary sinus ostium (Herman K, 1951). Thebesian valve was found to be present in majority of the hearts. Of these, 16% had a valve morphology covering greater than 75% of the ostium. It could be fibrous, fibromuscular, or muscular composition and devoid of fenestrations. These make them potentially complicating structures interfering with the cannulation of coronary sinus (Gary et al., 2009). In the present study 20 % were valveless, crescentic and membranous in 46% and in remaining fenestrations were present. Hence catheterization may be difficult in some of the cases.
Embryology
Coronary sinus is derived from the left horn and body of the sinus venosus. Valve of the coronary sinus is derived from lower part of right venous valve. Variations in the shape of the thebesian valve could be due to defective formation or absorption of lower end of the right venous valve.
Conclusion
Since in many specimens coronary sinus was accompanied by coronary arteries, the arteries may help in venous return by their pulsations. Anatomy of coronary valve and variations are important in catheterization of the coronary sinus. There are numerous variations of the coronary veins, coronary sinus, valves, all of which may affect the degree of myocardial protection provided by retrograde cardioplegia. More anatomical studies can be done using radiology to know the segmental drainage of the heart and coronary venous anatomy as it is relevant in cardiac resynchronization therapy and electrophysiological studies.
References
Abraham WT, Hayes DL (2003) Cardiac resynchronization therapy for heart failure. Circulation, 108, 2596-603.
Cappato R, Schulter M, Weiss C, et al. (1997) Mapping of the coronary sinus and great cardiac vein using a 2-French electrode catheter and a right femoral approach. J Cardiovasc Electro-physiol, 8, 371-6.
Gary SM, Alexander JH, Florin M, Krishnan SC (2009) Variations in Thebesian valve anatomy and coronary sinus ostium: implications for invasive electrophysiology procedures. Europace, 11: 1188-1192.
Gerber TC, Sheedy PF, Bell MR, et al (2001) Evaluation of the coronary venous system using electron beam computed tomography. Int J Cardiovasc Imaging, 17: 65-75.
Herman K, Hellerstein J, Lowell Orbison (1951) Anatomic Variations of the Orifice of the Human coronary Sinus. Circulation, 3: 514-523.
Praveen I (2006) Great Cardiac Vein draining directly into the Right Atrium -A Case Report. J Anat Soc India, 55: 60-64.
Jongbloed MR, Lamb HJ, Bax JJ, Schuijf JD, de Roos A, van der Wall EE, Schalij MJ (2005) Noninvasive Visualization of the Cardiac Venous System Using Multislice Computed Tomography. J Am Coll Cardiol, 45(5), 749-53.
Nico VR, Schuijf JD, De Sutter J, Devos D, Bleeker GB, de Roos A, van der Wall EE, Schalij MJ, Bax JJ (2006) Non-Invasive visualization of the Cardiac Venous System in coronary artery disease patients using 64-slice computed tomography. J Am Coll Cardiol, 48, 1832-1838.
Ortale JR, Gabriel EA, Lost C, Marquez CQ (2001) The anatomy of the coronary sinus and its tributaries. Surg Radiol Anat, 23(1), 15-21.
Permyos Ruengsakulrach, Buxton BF (2001) Anatomic and hemodynamic considerations influencing the efficiency of retrograde cardioplegia. Ann Thorac Surg, 71, 1389-1395.
Puglisi A, Lunati M, Marullo AG, et al. (2004) Limited thoracotomy as a second choice alternative to transvenous implant for cardiac resynchronization therapy delivery. Eur Heart J, 25, 1063-1069.
Ronald A, Bergman Adel K, Afifi Ryosuke Miyauchi (2010) Coronary Sinus, Illustrated Encyclopedia of Human Anatomic Variation: Opus II: Cardiovascu;ar System: Veins: Head, Neck, and Thorax (online). Available from http://www. anatomyatlases.org [9th July 2012]
Singh JP, Houser S, Heist EK, Ruskin JN (2005) The coronary venous anatomy: a segmental approach to aid cardiac resynchronization therapy. J Am Coll Cardiol, 46: 68-74
Susan Standring (ed) (2008) Gray’s Anatomy: The anatomical basis of clinical practice. Spain, Churchill Livingstone.